Proces spalania drewna
Dla poprawnego spalania potrzebne jest spełnienie warunków dobrego wymieszania paliwa z powietrzem oraz odpowiedniej temperatury w palenisku nie mniejszej od 600°C. Najłatwiej jest spalać gaz, gdyż wymieszanie gazu z powietrzem nie nastręcza żadnych kłopotów. Spalanie paliw stałych jest dużo trudniejsze, gdyż trudniejsze jest doprowadzenie cząstek powietrza do bezpośredniego kontaktu z cząstkami paliwa.
Proces spalania drewna odbywa się w trzech etapach:
| • | suszenie, |
| • | gazyfikacja i spalanie, |
| • | dopalanie się węgla drzewnego. |
Podczas podgrzewania drewna najpierw zachodzi proces odparowania wody oraz powierzchniowej gazyfikacji czyli rozpadu związków chemicznych pod wpływem odpowiednio wysokiej temperatury, tzw. piroliza. Po odparowaniu wilgoci proces ten przenosi się w głąb drewna. Gaz w kontakcie z powietrzem spala się płomieniem. Drewno charakteryzuje się bardzo dużą zawartością części lotnych wynoszącą do 80% (węgiel ok. 30%). Pozostałe 20% spala się jako żarzący się węgiel drzewny aż do całkowitego wypalenia za wyjątkiem związków niepalnych, które tworzą popiół.
Popiół drzewny uznawany jest za dobry nawóz rolniczy. Składa się on ze związków krzemu (Si) oraz potasu (K), sodu (Na), fosforu (P), wapnia (Ca) i magnezu (Mg). Ważną cechą popiołu jest jego temperatura topnienia, która obniża się wraz ze wzrostem zawartości potasu i częściowo sodu. Zbyt niska temperatura topnienia (poniżej 1050°C) może być przyczyną zanieczyszczeń wewnętrznych powierzchni kotła. problem ten występuje głównie przy spalaniu słomy, w której zawartość potasu może być nawet 10 razy większa niż w drewnie.
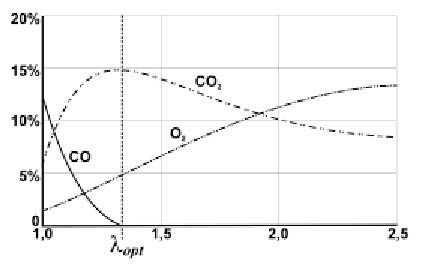
Obok temperatury w komorze spalania, która w każdym miejscu musi być wyższa od 700°C ważna dla procesu spalania jest ilość powietrza doprowadzana do kotła. Dla różnych palenisk i rodzajów drewna wymagane są różne wartości tzw. współczynnika nadmiaru powietrza L (lambda), określającego ile razy ilość powietrza jest większa od ilości teoretycznej wynikającej ze wzorów stechiometrycznych.
Za mała ilość powietrza powoduje niedopalenie się cząstek węgla i powstanie tlenku węgla, a także przedostawanie się do spalin niespalonych węglowodorów. Z kolei zbyt duża ilość powietrza powoduje wychładzanie kotła (część powietrza nie bierze udziału w spalaniu) i obniżenie sprawności, a także sprzyja powstawaniu szkodliwych tlenków azotu NOx.
Ponadto, duży nadmiar powietrza powoduje podwyższenie temperatury płomienia spalania, co z kolei przyczynia się do, niekorzystnego dla kotła, topnienia popiołu.
Orientacyjne wielkości współczynnika nadmiaru powietrza L przedstawiono w tabeli, a zależności pomiędzy lambda i procentowymi zawartościami O2 i CO2 na wykresie:
| współczynnik nadmiaru powietrza L |
O2 [%] |
|
| kominek, szczapy | 2,3 ÷ 3,0 | 12 ÷ 14 |
| piec na szczapy | 2,1 ÷ 3,0 | 11 ÷ 12 |
| kotły na zrębki | 1,4 ÷ 1,6 | 6,0 ÷ 8,0 |
| kotły na pelety | 1,2 ÷ 1,6 | 4,0 ÷ 8,0 |
Właściciel kotła lub kominka powinien sprawdzać od czasu do czasu parametry spalania umożliwiające oznaczenie sprawności urządzenia. Można to zrobić za pomocą własnych, prostych urządzeń do analizy spalin (np. pompka z chemicznym znacznikiem zawartości CO2 w spalinach) lub skorzystać z pomocy serwisu.
Trzeba przy tym wiedzieć, że:
| • | do spalenia 1 kg suchego drewna potrzeba 3,5-4 m³ powietrza, |
| • | maksymalna zawartość CO2 dla spalin CO2 max = 20,2 [%], |
| • | współczynnik nadmiaru powietrza L = CO2/ CO2 max, |
| • | zamienność CO2 i O2 dla drewna ustala się wg wzoru: O2 [%] = 1,04 (20,2 – CO2 [%]) |
| • | stratę wylotową, Swyl odczytuje się z wykresu znając zawartość O2 w spalinach i ich temperaturę na wyjściu z kotła: |
Sprawność kotła oblicza
się w przybliżeniu L = 100% – Swyl